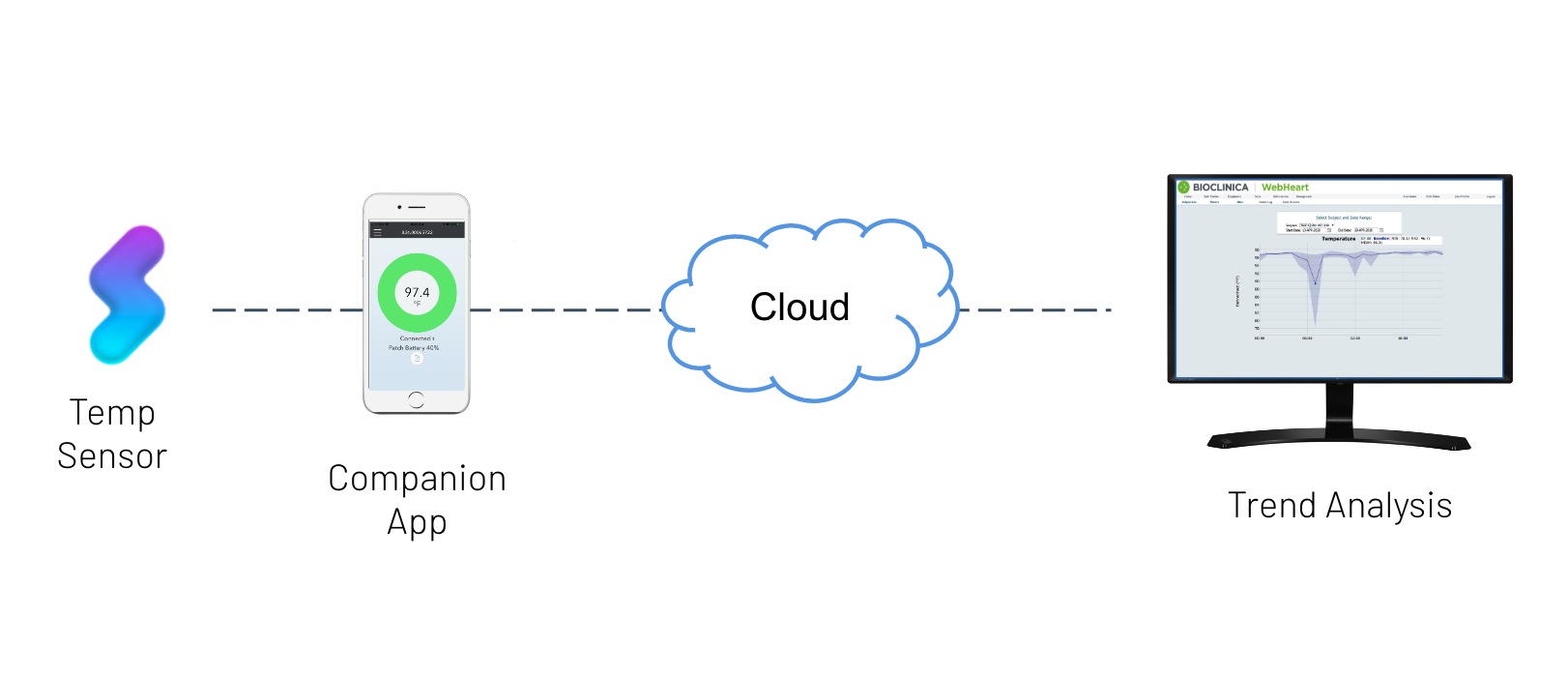
One of the primary indicators of patient safety and efficacy in this trial is changes in body temperature over extended periods.
This study leveraged Vivalink’s Biometric Data Platform the medical grade wearable temperature monitor to analyze patient temperature in ambulatory and at-home settings over a 3 week period.
| Requirements/Challenges | Solution |
| Ambulatory impacts to temperature variations | Trend analysis to flag artificial temperature changes |
| Extended monitoring period | 21 day rechargeable temperature sensors |
| Network disruptions in ambulatory settings | Sensors with onboard memory and automated network data synchronization |
| Data integration with clinical systems | M2M web services API integration |
"The integration of Vivalink's innovative RPM technology with Bioclinica's global clinical trial infrastructure will open new paradigms for trial data collection and analysis."
- Thomas Fuerst, PhD, Chief Science Officer of BioClinica
Copyright © 2026 VivaLNK, Inc. All Rights Reserved.