Enhance patient COPD care through home monitoring solutions. This multi-center observational study correlates wearable sensor data with clinical parameters to monitor COPD. It seeks to combine parameters such as heart rate, HRV, respiratory rate, and breathing patterns to gauge improvement or degradation of COPD.
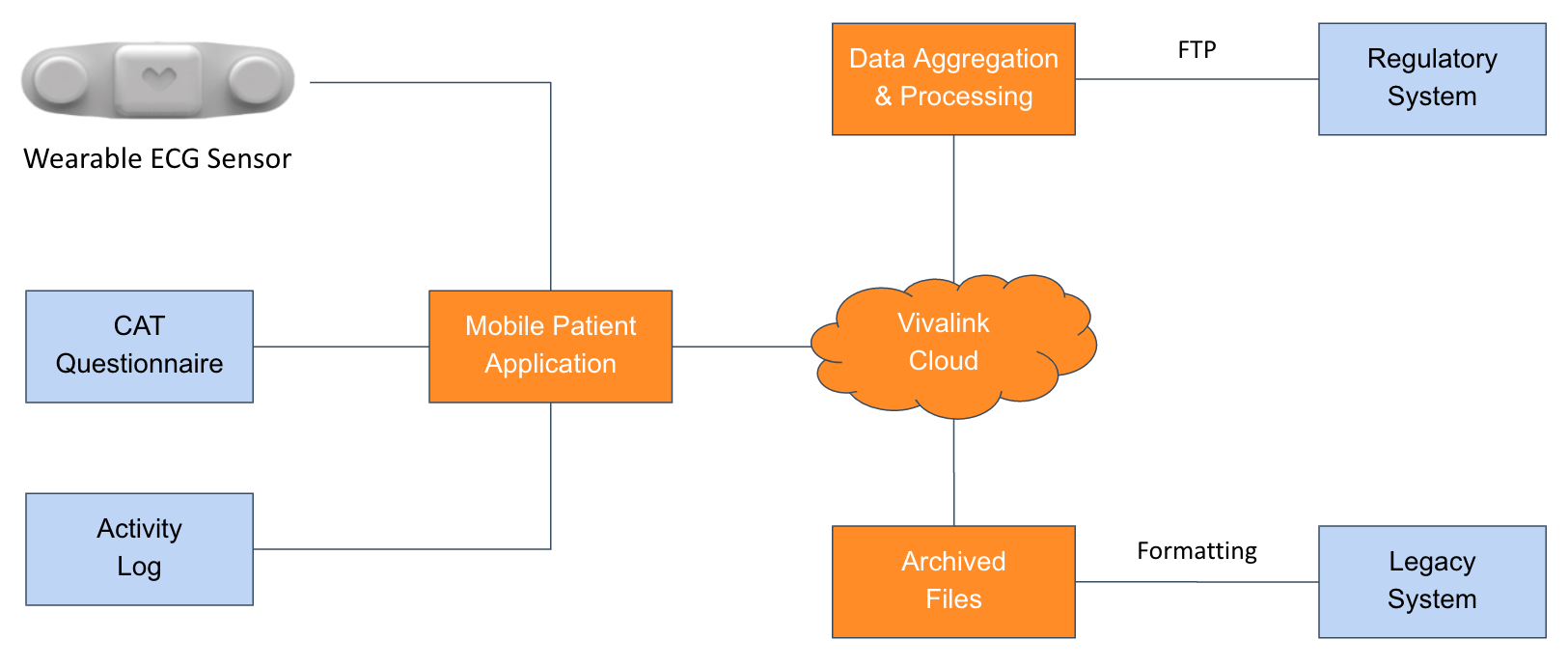
Participants provide data using a combination of wearable sensors and self-reported symptoms from remote, at-home locations. The data is then consolidated and processed centrally, integrated with regulatory systems, and formatted for archive into legacy systems.
| Requirements/Challenges | Solution |
| Complex clinical patient app requirements | Customized app with ePRO, surveys |
| Legacy data format and systems | Flexible data formats and integration options |
| Data privacy concerns outside of region | Regional data hosting in country |
| Patient language barriers | Local translation for patient facing content |
| Simplified onboarding experience | Turnkey RPM kits including cellular service |
| Ensuring patient adherence | Patient compliance dashboard |
Vivalink offers quality management system, device, software and is able to provide end-to-end support. [We are] confident that Vivalink can offer and maintain the highest standards of the service.
- Trial Manager
Copyright © 2026 VivaLNK, Inc. All Rights Reserved.