As drug development continues to advance, concern for patient safety remains a top priority. Many drugs can have negative effects on the heart’s rhythm and function, leading to serious cardiac events including arrhythmias, QT interval prolongation, or heart failure. Using cardiac safety monitoring during drug development both protects participants from harm and reduces the risk of late-stage trial failures, allowing potentially life-saving therapies to reach the market.
While traditional approaches to cardiac monitoring during drug trials serve their purpose, they can also pose a problem for drug developers. If the heart was like a leaky faucet, traditional methods would be the equivalent of checking it once a day. They rely on periodic, in-clinic electrocardiogram (ECGs), which can often miss arrhythmias or other drug-induced cardiac changes that occur outside those limited windows, creating problematic blind spots. Consequently, important signals of conditions like Atrial fibrillation (AFib) may go undetected.
A recent study conducted by Brigham and Women’s Hospital (BWH) sheds light on a solution for enhancing safety during drug trials. The study supported a way to enhance cardiac safety during drug trials in both enhancing signal detection and improving trial efficiency—through continuous, validated remote cardiac monitoring using medical-grade wearable ECGs.
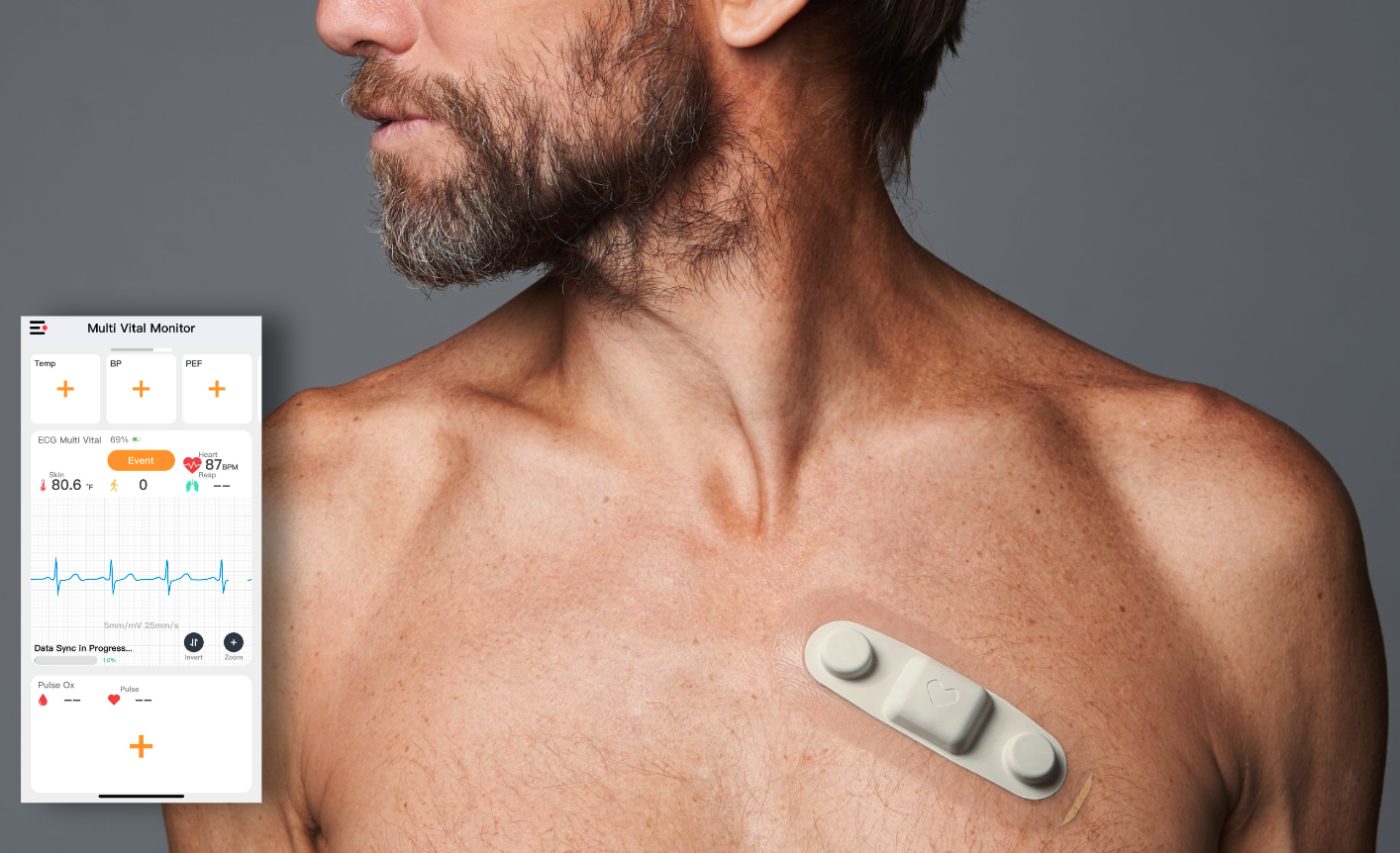
AFib is a common post-cardiac surgery complication, affecting up to 40% of patients. BWH researchers examined a gap in traditional patient monitoring: the possibility of missing AFib in patients once they left the hospital after cardiac surgery. Their study aimed to discover the consequences of no longer monitoring these patients once they leave the hospital, recording any post-discharge signs of AFib. They did this by enrolling over 100 cardiac surgery patients and equipped them with a Vivalink wearable ECG patch, which they wore for up to 14 days after discharge.
The results revealed AFib in 27% of patients post-discharge. What’s more, 24% of those patients with AFib never showed any sign of it during hospitalization. Many of these instances showed up as intermittent episodes that lasted less than five minutes, too quick to be reliably caught by standard in-hospital or clinic ECGs. If those same AFib events were happening during drug trials, clinicians would be missing important data and potential red flags. By catching these missed events by using wearables in studies, clinicians can ensure faster intervention, improved data capture, and significantly more successful drug trials.
Traditional methods that monitor heart activity are only able to capture brief snapshots of the heart. They fail to account for transient or asymptomatic events, which might indicate a drug-related safety risk. To ensure the safety of participants in drug trials, we need a more reliable monitoring method.
The study conducted by BWH revealed that wearable ECGs are able to capture those short-term arrhythmias normally missed by periodic snapshots. The devices’ enhanced signal detection ensures that any subtle or fleeting cardiac irregularities are caught sooner rather than later. This real-time visibility gives teams the chance to intervene as soon as a red flag is raised, allowing them to adjust the dose or stop the trial altogether.
Beyond ensuring patient safety, continuous wearable monitoring can reinforce regulatory confidence. Objective, real-world data is valued for safety assessments, and continuous monitoring provides just that – heart rhythm trends, heart rate variability, and the frequency and duration of arrhythmias. This data helps support stronger safety narratives, reducing the uncertainty of having missed something between visits.
If traditional cardiac safety monitoring is like checking a leaky faucet once a day, wearable ECGs are like installing a smart water meter that records each drop, at every moment of the day.
The study conducted by BWH is a powerful indicator of the advantages continuous, wearable ECG monitoring can provide. By uncovering clinically meaningful arrhythmias that standard care might miss, wearable ECGs can increase both patient safety and trial effectiveness. Using this technology during drug development can equip us with better signal detection, improved safety data, faster risk management, and stronger regulatory confidence.
Continuous, high-fidelity cardiac data can reveal clinically meaningful events that traditional monitoring overlooks. For sponsors and CROs, that translates into stronger safety profiles, fewer blind spots, and greater confidence in trial outcomes. To learn how Vivalink’s ECG patch can support cardiac safety monitoring in your upcoming studies, connect with our team.
Copyright © 2026 VivaLNK, Inc. All Rights Reserved.
No Comments Yet
Let us know what you think