The Six-Minute Walk Test (6MWT) is a practical, simple test that measures the distance a patient can quickly walk on a flat, hard surface in a span of six minutes This globally recognized evaluation involves all systems during exercise, from the pulmonary and cardiovascular systems to peripheral circulation, blood, neuromuscular units, and muscle metabolism. The test doesn't require exercise equipment or advanced training for patients making it easy for clinicians to administer.
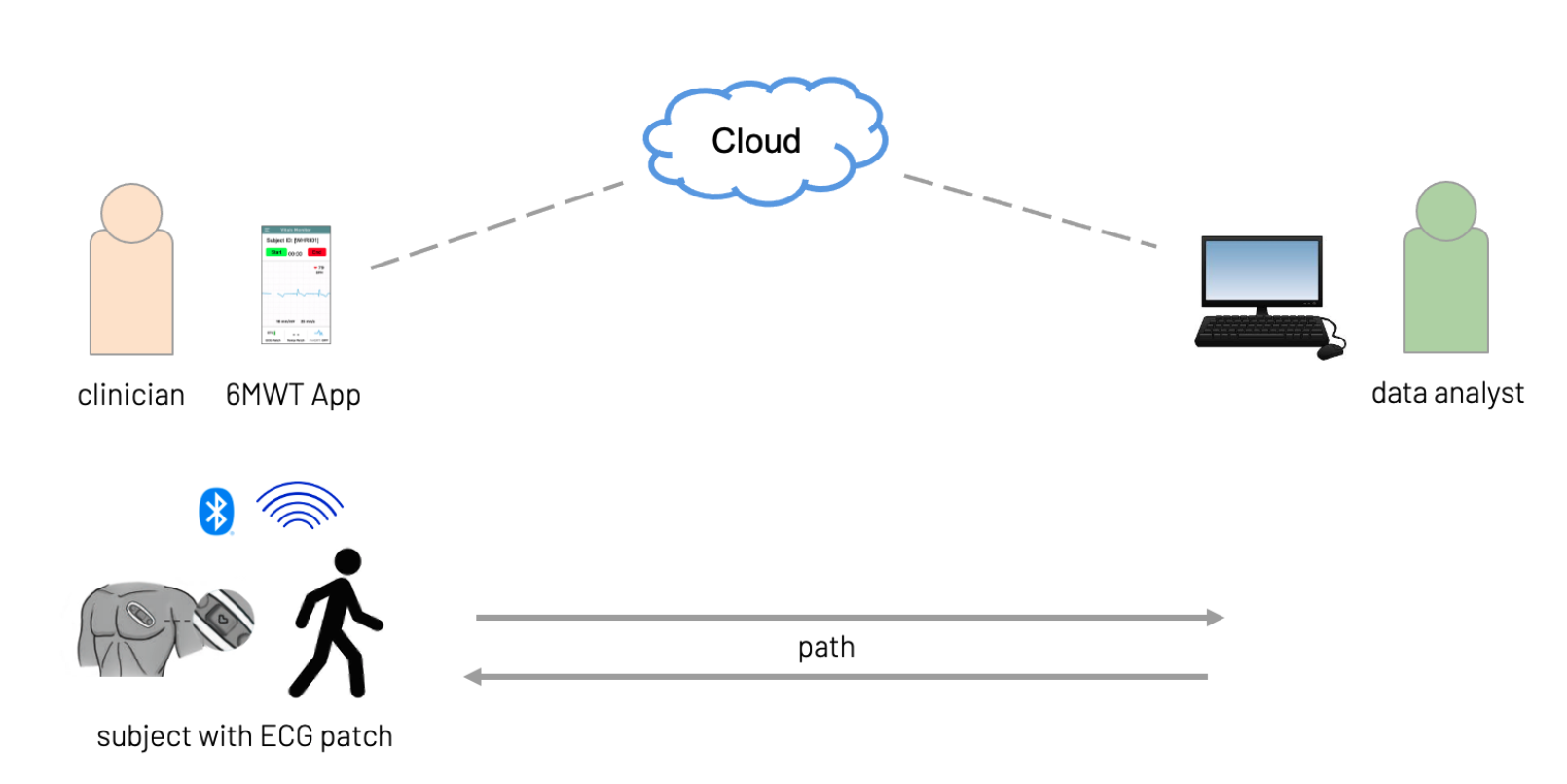
This multi-site, multi-country study involves a six-minute walk test protocol designed to capture ECG rhythm and heart rate before, during and after the routine, while the patient walks a 90 foot designated path.
This study leveraged Vivalink’s Biometric Data Platform and the wearable ECG monitor to analyze patient heart rate changes throughout the routine.
| Requirements/Challenges | Solution |
| Specific clinical protocol required to manage 6MWT routine | Customized clinical mobile app to match protocol use case |
| Logistical challenges with international sites | Provisioning and direct ship to international sites |
| Aggregating data from multi-sites | Consolidated data for centralized analysis |
| Network disconnections during 6MWT routine | Auto network recovery and data synchronization |
| Ensuring data adherence for RPM devices | Data acquisition reports to monitor adherence |
"Medical wearables are making it possible to explore new, potentially meaningful outcomes in clinical studies without adding much additional burden to patients or their clinicians."
-Robert F. Roscigno, PhD, Vice President, Clinical Development at Gossamer Bio
Copyright © 2026 VivaLNK, Inc. All Rights Reserved.